Calculate Energy Required to Boil Water in the Two-Phase Fluid Domain
This example shows how to calculate the amount of energy needed to lift the lid of a container filled with water by boiling the water. First, you solve for the solution analytically, and then build a Simscape™ model to represent the physical scenario and validate your results.
Solve for Energy Needed to Lift the Lid Analytically
Consider a sealed pot of water at room temperature that has 10 kg of weight on the lid. The radius of the pot is 15 cm and the height is 10 cm. The pot is on a heat source that is 400 K. Ignore the effects of the pot wall. The pot is initially at atmospheric pressure and pot is completely filled with liquid water.
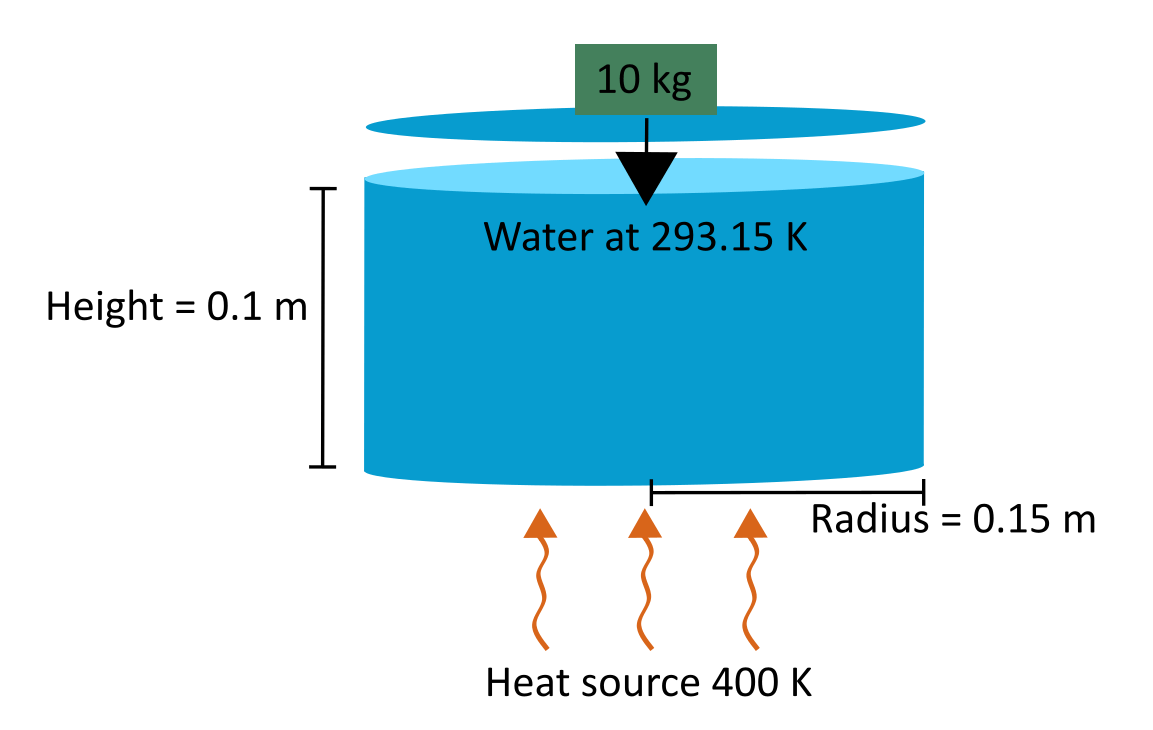
Create MATLAB® parameters to represent the pot temperature, dimensions, and water characteristics.
r_pot = 0.15; % [m] Pot radius h_pot = 0.1; % [m] Pot height T_boil = 373.15; % [K] Water boiling temperature T_room = 293.14; % [K] Water room temperature m_lid = 10; % [kg] Mass of lid M = 0.018; % [kg/mol] Molar mass of water c = 4186; % [J/kg*K] Specific heat capacity of water L = 2.26 * 10^6; % [J/kg] Latent heat of vaporization of water R = 461.52; % [J/(kg*K)] Water vapor gas constant
Calculate the pot volume, surface area, force pushing down on the lid, and mass of water in the pot.
SA = r_pot^2 * pi; % [m^2] Surface area of lid V_pot = h_pot * SA; % [m^3] Pot volume F_lid = m_lid * 9.81; % [N] Force of gravity on the lid m_w = V_pot * 997; % [kg] Mass of water
Calculate the pressure from gravity on the lid, which is the force of gravity acting on the lid divided by the lid surface area. This value is the pressure of fluid acting on the lid necessary to overcome the force of gravity.
p_lid = F_lid / SA; % [Pa] Pressure of fluid acting on the lid necessary to overcome the force of gravity The total energy required to lift the lid is the sum of the energy required to bring the entire volume of water to a boil and the energy required to vaporize some portion of that water. The energy required to bring the volume of water to boil is proportional to the temperature difference between room temperature and the boiling point,
where:
is the mass of the water.
is the specific heat capacity of water.
and are the boiling temperature and room temperature, respectively.
Use the ideal gas law to calculate how much of the water needs to vaporize to overcome the pressure due to the gravity force acting on the lid. The ideal gas law states
where:
is the pressure. This value is the fluid pressure required to overcome the force of gravity and lift the lid.
is the volume of the water vapor.
is the number of moles of water vapor.
is the gas constant of water vapor.
is the temperature of the water vapor. Because the lid lifts soon after the water boils, assume that the temperature is equal to the boiling temperature.
Because , you can use the molar mass of water, , to rewrite the equation in terms of the mass of the vapor,
To calculate the vapor volume, assume the lid lifts after a small displacement of 0.5 cm. Because the energy needed to vaporize the water is negligible compared to , this assumption does not impact the final solution. Use this value to calculate the mass of the water vapor.
V_vapor = 0.005 * SA; % [m^3] Volume of the vapor m_v = (V_vapor * M * p_lid)/(R*T_boil); % [kg] Mass of water vapor
The energy needed to vaporize this mass of water is
where is the latent heat of vaporization of water.
Use these values to calculate the total energy required to move the lid,
E = m_w * c * (T_boil - T_room) + m_v * L % [J] Total energy requiredE = 2.3603e+06
Build the Model
Next, build a model that represents the physical system. To represent this scenario, the model needs three domains. The two-phase fluid domain models the boiling water, the thermal domain models the heat source, and the mechanical domain models the pot lid and the forces acting on it.
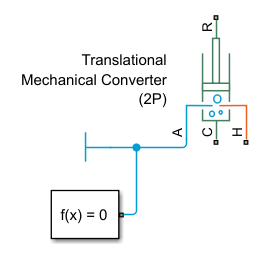
Add Two-Phase Fluid Blocks
Add a Translational Mechanical Converter (2P) block. This block models the pot and the pressure it exerts on the lid, which is represented by the converter interface.
Set the block parameters of the Translational Mechanical Converter (2P) block to match the physical scenario. Set the values of the Initial interface displacement parameter to
h_pot mand the value of the value of the Interface cross-sectional area parameter toSA m^2.Connect port A of the block to a Cap (2P) block, which terminates the network and models the liquid in the pot as sealed.
Add and connect a Solver Configuration block.
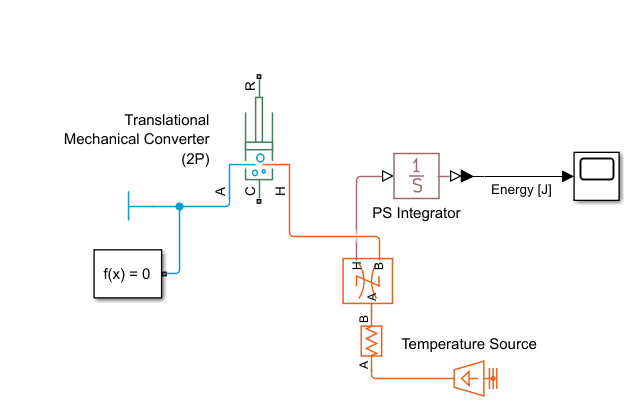
Add Thermal Blocks
To represent a heat source, add a Temperature Source block to the model. Set the Temperature parameter to
400 K.Connect the Temperature Source block to port H of the Translational Mechanical Converter (2P) block.
To maintain numerical stability, add a Thermal Resistance block between the temperature source and the converter. Set the Thermal resistance parameter to a negligible value,
0.001 K/W.
These three blocks model the physical effects of the heating pot. Next, add blocks that calculate the energy used and display the results.
Add a Heat Flow Rate Sensor block between the Thermal Resistance and the converter.
The Heat Flow Rate Sensor block only measures the heat flow rate, or power, into the pot. Energy is the integral of power. To calculate the energy applied to the pot, connect a PS Integrator block to the sensor block port H.
To specify the integral initial conditions, set the PS Integrator block Initial condition parameter to
0 J.Connect the PS Integrator output to a PS-Simulink Converter and a Scope block.
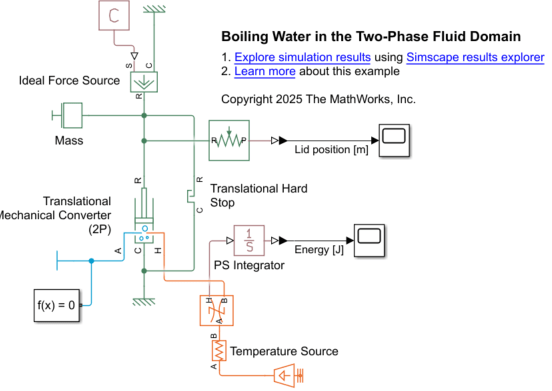
Add Mechanical Blocks
Connect a Mechanical Translational Reference block to port R of the Translational Mechanical Converter (2P) block.
To represent the force of gravity acting on the lid, add a Ideal Force Source block and connect port R to port R of the converter block. Connect a Mechanical Translational Reference block to port C of the Ideal Force Source block.
Connect a PS Constant block to the force source. Set the Constant parameter to
F_lid N.Connect a Translational Hard Stop block in parallel with the converter block. The hard stop represents the pot casing, which prevents the lid from falling through the top of the pot. When fluid pressure is less than the required pressure to overcome gravity, the hard-stop force counteracts the force of gravity acting on the lid.
In the hard stop block, set the Upper bound and the Lower bound parameters to
infandh_pot m, respectively. To reduce the numerical impact of damping and stiffness at the lower bound, set Hard stop model toFull stiffness and damping applied at bounds, undamped rebound, Contact stiffness at lower bound to1e2 N/m, and Contact damping at lower bound to50 N*s/m.Add a Mass block to represent the lid. The mass accelerates when there is an imbalance between the force of gravity, fluid force, and hard stop force. Set the Mass parameter to
m_lid kg.To measure the pot lid position, add an Ideal Translational Motion Sensor block. Set Measurement reference to
Differenceand Initial position toh_pot m. Connect the sensor block port R to the signal leaving port R of the Translational Mechanical Converter (2P) block. Connect the output to a PS-Simulink Converter and a Scope block.
Open the BoilingWaterin2PDomain model to see the completed model.
Compare Results
Simulate the BoilingWaterin2PDomain model and plot the temperature of the two-phase fluid.
mdl = "BoilingWaterin2PDomain"; open_system(mdl); sim(mdl); t_model = tout; % [sec] Model time temp = simlog_BoilingWaterin2PDomain.Translational_Mechanical_Converter_2P.T_I.series.values; % [K] Temperature plot(t_model, temp) xlabel("Time (s)") ylabel("Water Temperature (K)")
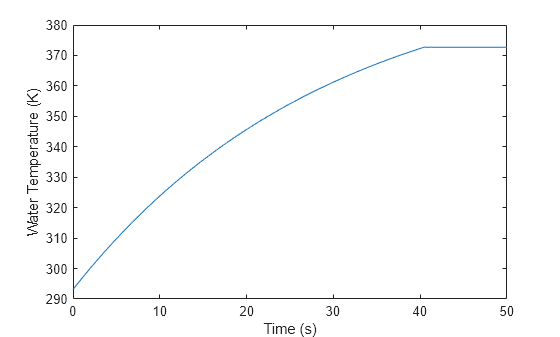
From 0 to approximately 40 seconds, the water temperature rises as it approaches the boiling point. Around 40 seconds, the temperature becomes constant as the water vaporizes. After the water starts to vaporize, the translational mechanical converter interface, which represents the pot lid, can move.
View the position of the lid by opening the Scope block connected to the Lid position [m] signal.
open_system(mdl + "/Scope1")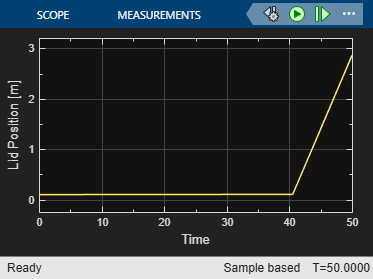
The lid begins to move at approximately 40 seconds. Find the time the lid begins to move above the initial displacement.
position = simlog_BoilingWaterin2PDomain.Translational_Mechanical_Converter_2P.interface_displacement.series.values; % [m] Lid position f = find(position > (h_pot +.005), 1); % Index of position array where lid moves past initial displacement time_move = t_model(f) % [s] Time that lid moves
time_move = 40.4643
The lid begins to move at approximately 40.47 seconds. Find the total energy used at the time the lid begins to move.
Energy = simlog_BoilingWaterin2PDomain.PS_Integrator.x.series.values; % [J] Energy applied to pot over simulation time Energy_move = Energy(f) % [J] Energy value when lid moves
Energy_move = 2.3559e+06
The models results show it takes about 2,356.2 kJ to move the pot lid, which agrees with the calculated value.
References
[1] Adrienne S. Lavine, Frank P. Incropera, David P. DeWitt, Theodore L. Bergman. Fundamentals of heat and mass transfer. Vol. 6. New York: Wiley, 1996.